Gain an understanding of disease pathogenesis and identify novel therapeutic targets with our full-spectrum, non-blood human biospecimens — including Cerebralspinal fluid and respiratory swabs.
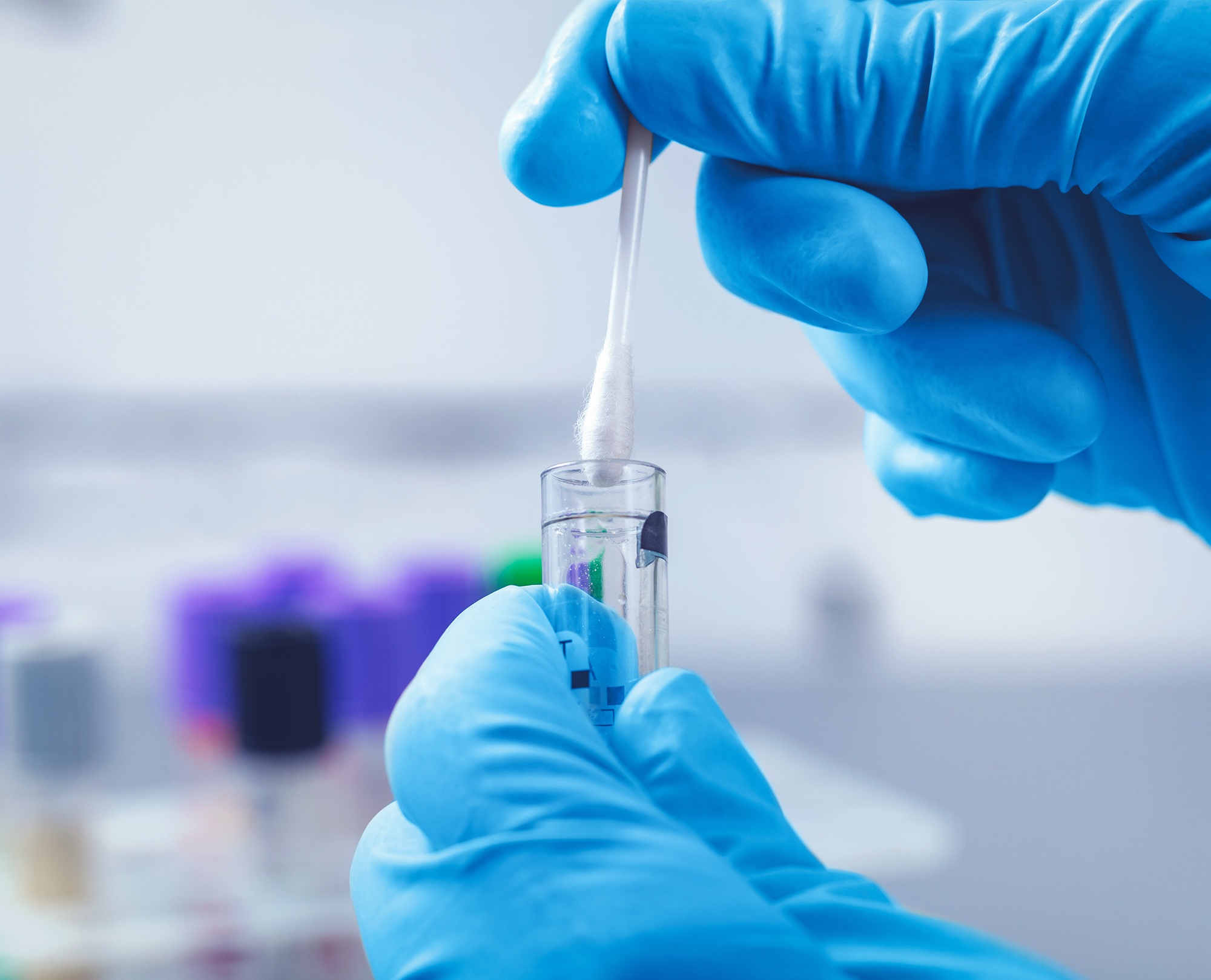
Diagnostic remnant samples available across a range of demographic characteristics.
Remnant samples from multiple respiratory viral panels are separated by positive and negative results
Sputum samples from multiple diagnostic tests available with both positives and negatives.
Culture positive endotracheal aspirates across a range of demographic characteristics.
Get biospecimens in the format you need – then deeply characterize them with Discovery’s customizable and scalable biomarker services based on advanced technologies, including genomics, proteomics, molecular pathology, and cell biology services.